The discovery and validation of Panx1 opened a transformative pathway for developing selective blockers, offering new hope in treating cancers and neurological disorders. Pannex Therapeutics drives this innovation with its drug discovery platform.
Panx1 is an ATP-releasing membrane channel, highly relevant in neuroinflammation and immune signaling.
Neurology
Chronic neuropathic pain
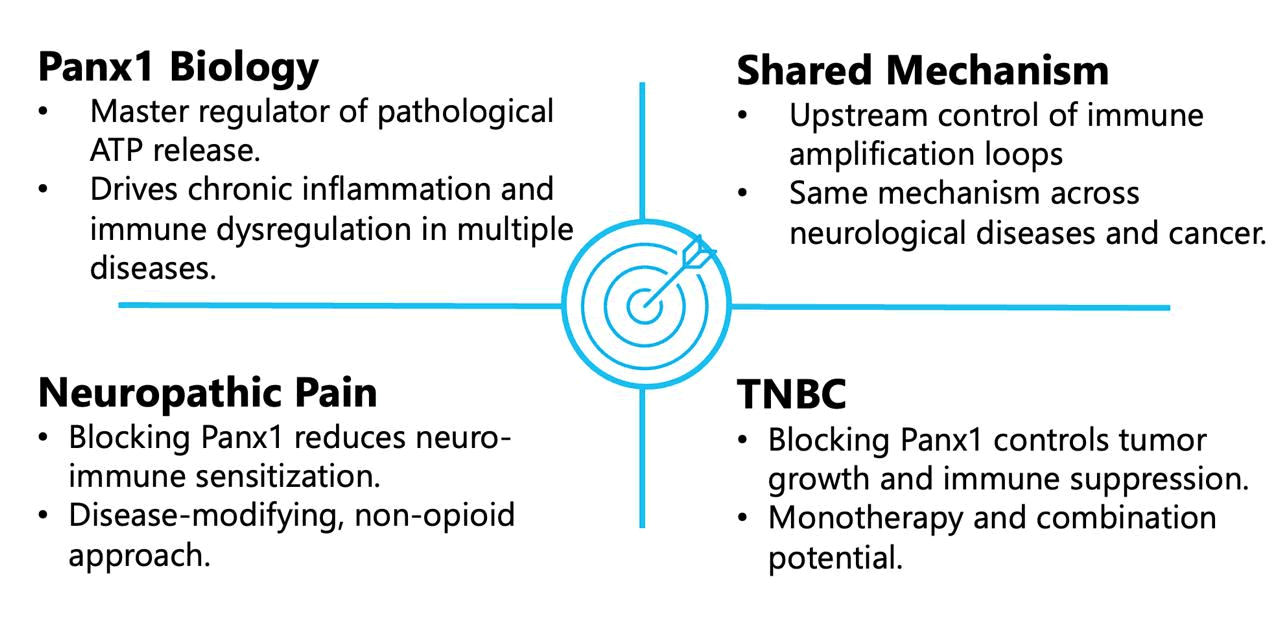
Pannexin 1 (Panx1) channels play a central role in pathological signaling in the nervous system by mediating stress-induced ATP release, a key trigger of neuroinflammation and abnormal neuron–glia communication. Under conditions of nerve injury or persistent tissue damage, Panx1 becomes aberrantly activated in neurons, glial cells, and immune cells, initiating and sustaining inflammatory signaling cascades. In chronic neuropathic pain, this Panx1-driven ATP release amplifies microglial and astrocyte activation, promotes cytokine release, and reinforces central and peripheral sensitization of pain pathways. Because Panx1 sits upstream of these self-perpetuating immune and inflammatory loops, blocking the channel has the potential not only to reduce pain symptoms but to modify the underlying disease mechanisms that maintain chronic neuropathic pain.
Oncology
Triple negative breast cancer
Pannexin 1 (Panx1) channels play an important role in cancer biology by regulating pathological ATP release within the tumor microenvironment, where extracellular ATP acts as a powerful modulator of inflammation, immune responses, and tumor cell behavior. In oncology, aberrant Panx1 activity contributes to chronic immune dysregulation, supports pro-tumorigenic signaling, and shapes interactions between cancer cells and immune and stromal cells. In triple-negative breast cancer (TNBC), Panx1 has been implicated in promoting tumor growth, invasiveness, and resistance to stress through ATP-driven signaling pathways that enhance survival and immune evasion. By sitting upstream of these interconnected processes, Panx1 represents a compelling therapeutic target in TNBC, with the potential to disrupt tumor-supportive signaling networks and sensitize tumors to immune-mediated control.
Selected scientific publications
Panx1 in cancer:
1. Laird, D. W., & Penuela, S. (2021). Pannexin biology and emerging linkages to cancer. Trends in Cancer, 7(12), 1119-1131.
2. Jalaleddine, N., El-Hajjar, L., Dakik, H., Shaito, A., Saliba, J., Safi, R., … & El-Sabban, M. (2019). Pannexin1 is associated with enhanced epithelial-to-mesenchymal transition in human patient breast cancer tissues and in breast cancer cell lines. Cancers, 11(12), 1967.
3. Furlow, P. W., Zhang, S., Soong, T. D., Halberg, N., Goodarzi, H., Mangrum, C., … & Tavazoie, S. F. (2015). Mechanosensitive pannexin-1 channels mediate microvascular metastatic cell survival. Nature cell biology, 17(7), 943-952.
4. Chen, Wuzhen, et al. “High PANX1 Expression Leads to Neutrophil Recruitment and the Formation of a High Adenosine Immunosuppressive Tumor Microenvironment in Basal-like Breast Cancer.” Cancers 14.14 (2022): 3369.
5. Bao L, Sun K, Zhang X. PANX1 is a potential prognostic biomarker associated with immune infiltration in pancreatic adenocarcinoma: A pan-cancer analysis. Channels (Austin). 2021 Dec;15(1):680-696.
6. Shi G, Liu C, Yang Y, Song L, Liu X, Wang C, Peng Z, Li H, Zhong L. Panx1 promotes invasion-metastasis cascade in hepatocellularcarcinoma. J Cancer. 2019 Sep 7;10(23):5681-5688.
Panx1 in general:
1. Navis KE, Fan CY, Trang T, Thompson RJ, Derksen DJ. Pannexin 1 Channels as a Therapeutic Target: Structure, Inhibition, and Outlook. ACS Chem Neurosci. 2020 Aug 5;11(15):2163-2172. doi: 10.1021/acschemneuro.0c00333. Epub 2020 Jul 20. PMID: 32639715.
2. Sanchez-Arias JC, van der Slagt E, Vecchiarelli HA, Candlish RC, York N, Young PA, Shevtsova O, Juma A, Tremblay MÈ, Swayne LA. Purinergic signaling in nervous system health and disease: Focus on pannexin 1. Pharmacol Ther. 2021 Sep;225:107840.
3. Bhat EA, Sajjad N. Human Pannexin 1 channel: Insight in structure-function mechanism and its potential physiological roles. Mol Cell Biochem. 2021 Mar;476(3):1529-1540.